
The elements marked with an asterisk (in the 2nd column) have no stable nuclides.Lanthanoids and Actinoids are numbered as 101 and 102 to separate them in sorting by group. Group: There are only 18 groups in the periodic table that constitute the columns of the table.Elemental compositions of crustal rocks differ between different localities ( see article). Earth crust composition average values are from a report by F.In a sorted list, these elements are shown before other elements that have boiling points >0☌. The density of elements with boiling points below 0☌ is given in g/l.Periodic table 1 to 30 Element with Symbolsįollowing is the periodic table 1 to 30 elements with symbol.List of Periodic Table elements sorted by → Atomic number No. Hence, Atomic mass = Mass of protons + Mass of neutrons + Mass of electrons The atomic mass formula is shown below.Ītomic mass is the sum of protons, neutrons, and electrons. The mass of an atom is not expressed in units. This means that one atom has an atomic mass of exactly 12. The average mass of the naturally occurring isotopes of an element in comparison to the mass of an atom of 12C is known as relative atomic mass. Because electrons have a very low mass compared to protons or neutrons, their mass has no bearing on the calculation. 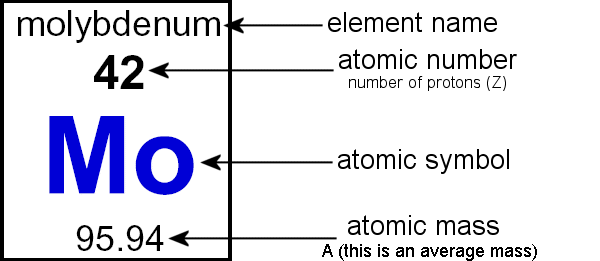
The sum of the masses of protons, neutrons, and electrons in an atom or group of atoms is called atomic mass. Overall atomic masses are determined for naturally occurring isotopes of each element, weighted by the weight of those individual isotopes on Earth, as shown in periodic tables like the one for hydrogen. The amount of example an isotope makes up determines how much it contributes to the normal. The normal of the atomic masses of the seeming plurality of distinct isotopes in an example is the general atomic mass. Because the isotopes of an element have different atomic masses, researchers can determine the element’s overall atomic mass (also known as the atomic weight). In most cases, the atomic mass number is rounded to the next whole number. Carbon-12, for example, is a typical carbon atom with six neutrons and six protons. The absolute mass of a single atom is its atomic mass, which is measured in atomic mass units, or amu. Carbon-12 has 6 protons and 6 neutrons, carbon-13 has 6 protons and 7 neutrons, and carbon-14 has 6 protons and 8 neutrons. For example, carbon has three isotopes: carbon-12, carbon-13, and carbon-14. Isotopes are atoms of the same element that have different numbers of neutrons. It is typically expressed in atomic mass units (amu), which are defined as 1/12 of the mass of a carbon-12 atom.The atomic mass of an atom is a weighted average of the masses of the isotopes of that atom. These are the first 30 elements in the periodic table, listed in order of increasing atomic number.Ītomic mass is the mass of an atom. Here is the periodic table of elements from 1 to 30: They can quickly solve problems based on atomic number and atomic mass in their board exams. Period Table 1 to 30 is very useful for the students of class 10. 
Periodic Table 1 to 30 Element with Symbol PDF. 
Atomic Mass of Elements 1 to 20 with Symbols.periodic table 1 to 30 Element with Symbols.NCERT Solutions Class 10 Social Science.NCERT Solutions For Statistics Class 11.
0 Comments
Leave a Reply. |
Details
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
